Introduction
Injection moulding enables the production of diverse disposable medical products, diagnostic equipment, and healthcare plastic components. This manufacturing method plays a critical role in medical device production for global markets by meeting stringent tolerance control, repeatable quality assurance, and product regulatory requirements, making it essential to the healthcare industry.
This article explores the application of injection moulding technology in medical device manufacturing, covering its advantages, types, processes, materials, standards, and latest advancements to assist you in selecting the appropriate manufacturing method.
What Is Medical Injection Moulding?
Medical injection moulding is a forming method that uses specialized equipment to manufacture plastic components for medical devices. It facilitates the production of specific parts while ensuring consistency and precision. Medical devices produced via injection moulding must guarantee zero functional defects to earn patient trust, while also enabling high-volume component manufacturing with precisely scheduled delivery cycles.

What Is the Medical Injection Moulding Process?
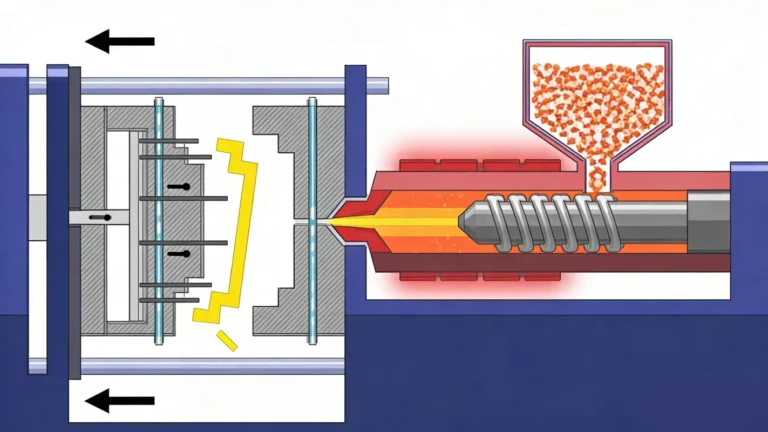
Medical injection moulding comprises five steps: feeding, melting, injection, cooling and ejection.
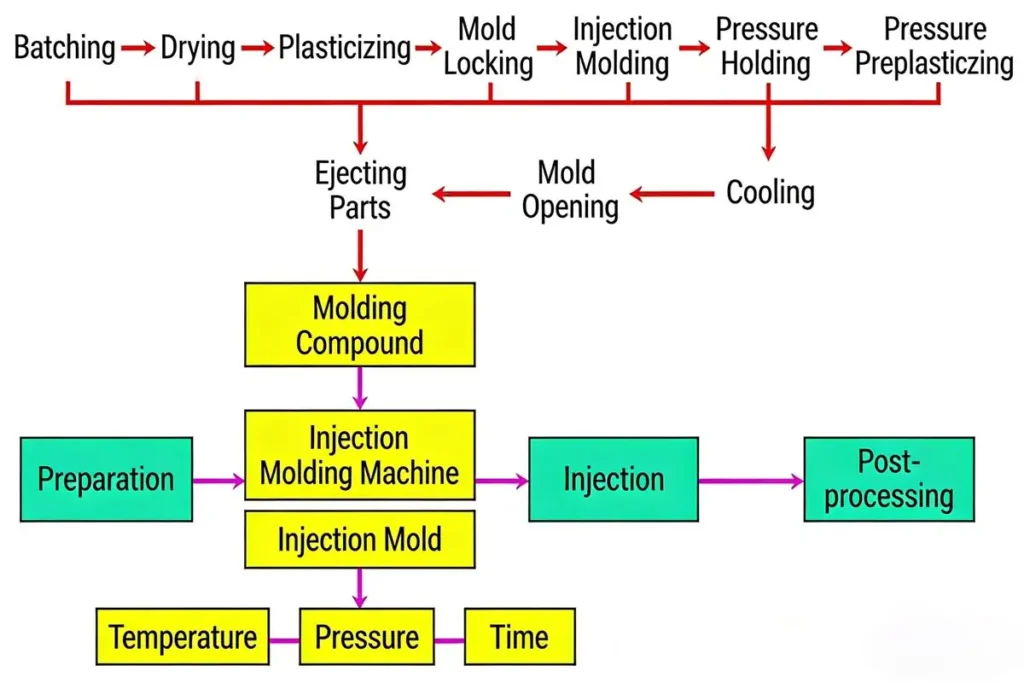
- Feeding: The first step involves loading typically medical-grade plastic pellets.
- Melting: Plastic pellets are heated within the injection moulding machine until they melt into a viscous liquid, ready for molding.
- Injection: The molten plastic is injected into the mold cavity under high pressure.
- Cooling: The molten material solidifies within the mold cavity, forming the perfect cavity shape.
- Ejection: After the mold opens, the part is ejected.
Why Has Injection Moulding Become the Mainstream Process for Medical Device Manufacturing?
Injection moulding is widely adopted in medical device production due to its high precision, consistent quality, low per-unit cos and versatile material options.
- Precision: Capable of achieving tight tolerance specifications, ensuring precise component fit and successful assembly.
- Quality: Once the mold is finalized, injection moulding enables consistent batch production with uniform quality.
- Cost: Large-scale production spreads mold costs, significantly reducing per-unit expenses.
- Materials: Diverse material options allow selection based on specific performance requirements.
- Structure: Complex parts can be produced as single, integrated components through mold design.
- Cleanrooms: Manufacturing in cleanroom environments minimizes contamination risks, meeting stringent hygiene and sterility requirements for medical devices.

What Injection Moulding Techniques Are Used in Medical Device Manufacturing?
The distinct characteristics, usage requirements, and intricate details of medical devices necessitate different plastic injection technologies. Currently employed techniques include insert molding, two-shot moulding, micro-injection moulding, thin-wall injection moulding, gas-assisted injection moulding, and liquid silicone rubber (LSR) injection moulding.

Insert Molding: Pre-placed components (e.g., metal inserts) are positioned in the mold cavity before injection. The molten material encapsulates the insert and solidifies during molding. Commonly used for precision instruments like metal tips on surgical tools.
Two-Shot Molding: First produces a base material, then overmolds with a soft plastic layer. This technique creates multi-material components, enhances grip comfort, and is widely used for medical device handles.
Micro-injection moulding: Utilizes miniature molds to produce intricate micro-components, commonly found in precision devices like hearing aids and microfluidic chips.
Thin-wall injection moulding: Enables production of medical components with wall thicknesses under 1mm, achieving material reduction and lightweighting. Suitable for needle holders, catheter housings, and similar applications.
Gas-Assisted injection moulding: Typically involves injecting inert gas into the mold cavity to form hollow runners. Commonly used for manufacturing device housings, surgical handles, and similar components.
Liquid Silicone Rubber injection moulding: Used to produce soft, elastic seals, tubing and other components. Safe for direct human contact applications.
What Types of Medical Products Are Produced Using Injection Moulding Technology?
Injection moulding technology is widely used in medical product manufacturing due to its ability to produce large volumes with consistent quality and high precision. Its applications in the medical supplies industry can be analyzed from three perspectives: disposable medical supplies, diagnostic and laboratory equipment, and implantable products.
Disposable Medical Devices
Leveraging its advantages of high-volume production, consistent quality, precision and low unit costs, injection moulding is employed to manufacture disposable medical devices with high consumption rates and moderate quality requirements, such as disposable syringes, sampling tubes and medical consumables.
Diagnostic And Laboratory Equipment
Injection moulding is employed for diagnostic and
laboratory equipment—such as IVD components, testing consumables and laboratory plastic parts—due to its ability to achieve precise dimensions, stable production cycles and high chemical resistance and safety in certain materials.
Implantable Products
Implantable products require prolonged contact with the human body, demanding materials with exceptional biocompatibility and sterility. Plastic injection precisely controls part geometry and surface finish, enabling consistent high-quality production. The resulting products are robust yet lightweight, offering excellent biocompatibility and sterility that fully meet medical requirements. Consequently, injection moulding is also employed for these medical products.
What Materials Are Commonly Used in Medical Injection Moulding?
There are approximately a dozen commonly used medical-grade plastics, including polyethylene (PE), polypropylene (PP), polyvinyl chloride (PVC), and polyurethane (PU). PVC and PE account for the largest shares at 28% and 24% respectively, PS represents 18%, PP constitutes 16%, and engineering plastics make up 14%.
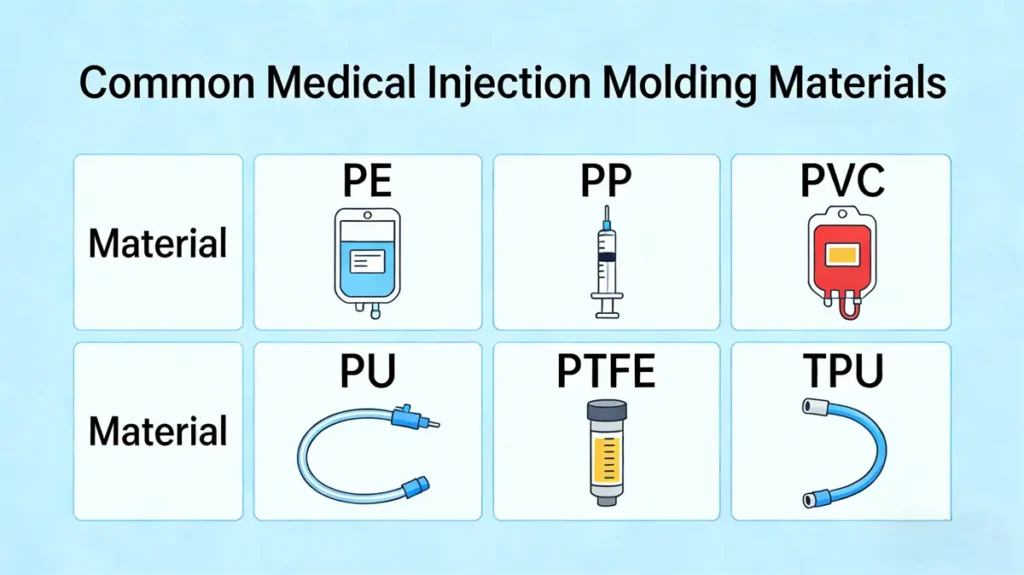
The selection of medical injection moulding materials often varies by region. For instance, in the United States and Europe, high-end medical devices typically utilize PEEK and PSU materials, while in emerging markets, disposable medical products widely employ cost-effective materials like PP and PVC.
1. PVC
PVC is a low-cost amorphous plastic with excellent chemical resistance and antimicrobial properties. Market estimates indicate PVC holds 28% of the medical plastic market.
2. PE
PE is the highest-volume general-purpose plastic due to its low cost and good processability. It can be rendered non-toxic, odorless, and biocompatible, commonly used for medicine bottles, needle caps, and syringe packaging bags.
3. PP
PP materials offer excellent chemical resistance, heat resistance, and processability. They feature high transparency, good barrier properties, and radiation resistance. Common applications include IV bags, disposable syringes, connectors, enteral nutrition packaging, and dialysis membranes.
4. PC
PC is characterized by high toughness, strength, and resistance to steam sterilization. Consequently, PC is the preferred material for blood dialysis filters, surgical instrument handles, and oxygen cylinders.
6. PTFE
PTFE, a fluoroplastic, possesses the lowest known friction coefficient among all plastics and superior chemical resistance. It offers excellent biocompatibility and anticoagulant properties, does not degrade, causes no adverse reactions when implanted in the human body, exhibits minimal aging, and has exceptional heat resistance, allowing for high-temperature sterilization.
It is widely used in the medical field for various artificial tracheostomies, artificial skin, artificial bones, and similar applications.
7. TPU
Thermoplastic elastomers exhibit outstanding low-temperature flexibility, hydrolysis resistance, anticoagulant properties, and resistance to microbial attack. They are commonly used in medical catheters, artificial hearts, oxygen masks, and similar applications.
What Are the Standards for Medical Injection Moulding?
Medical injection moulding is a manufacturing process with extremely stringent product requirements. The following are specific standards and relevant ISO certifications for medical injection moulding production. These standards are critical for medical devices supplied to markets in the United States, the European Union, and the Asia-Pacific region.
Specific Production Requirements
Medical injection moulding is a manufacturing process demanding high precision, cleanliness, and biosafety, directly impacting patient health and product compliance. Specific requirements can be categorized into four modules: materials, production environment, molds, and processes.
1. Materials
Material Selection: Must use FDA, CE, or NMPA-certified medical-grade materials such as polypropylene (PP), polycarbonate (PC), polyether ether ketone (PEEK), etc. Industrial-grade or recycled materials are strictly prohibited.
Biocompatibility: Select materials with corresponding biocompatibility grades based on product contact type. Must pass ISO 10993 biocompatibility testing.
Performance Suitability: Evaluate material properties including transparency, chemical resistance, impact resistance and high-temperature sterilization tolerance.
2. Production Environment
Cleanliness: Standard consumables require ISO Class 8 cleanrooms; implantable medical devices demand Class 10,000 or higher.
Environmental Control: Strictly regulate temperature (18-26°C), humidity (45%-65%), and pressure differentials. Conduct regular environmental monitoring.
Contamination Control: Utilize oil-free injection moulding machines and automated part retrieval systems to minimize human contact and eliminate oil mist contamination risks.
3. Molds
Precision: Dimensional tolerances must be controlled within ±0.005mm, with surface roughness below 0.1μm
Structure: Employ high-precision machining techniques for complex features like micro-pores, threads, and thin walls
Ventilation: Vent groove depth controlled at 0.02-0.05mm to prevent scorching or silver streaks
Maintenance: Thorough cleaning and disinfection of molds before each injection cycle; use medical-grade release agents
4. Process
Temperature: Set according to material properties; excessive heat causes degradation, insufficient heat reduces flowability
Pressure and Speed: Employ multi-stage injection to avoid turbulence; typical injection pressure ranges from 80-150MPa
Hold Pressure & Cooling: Hold time must cover the melt shrinkage phase; cooling time ensures complete product solidification
Drying: Hygroscopic materials require 4-6 hours drying at 80-120°C to prevent injection moulding bubbles
ISO Certification
ISO certifications relevant to medical injection moulding primarily include ISO 13485 Quality Management System certification, ISO 10993 Biocompatibility testing and ISO 14644 Cleanrooms and Associated Controlled Environments.
ISO 13485 Quality Management System certification is the quality management standard for the medical device industry. It strictly monitors injection moulding process parameters such as temperature, pressure, and cleanliness to ensure product consistency.
ISO 10993 biocompatibility testing is the biological evaluation standard for medical devices, conducting safety tests on materials that come into contact with the human body.
ISO 14644 Cleanrooms and Related Controlled Environments is the internationally recognized standard system for cleanrooms and related environments, providing a comprehensive technical framework for the design, construction, operation, and maintenance of cleanrooms.
How Does Medical Device Classification Impact Injection Moulding?
Medical devices can be classified into Class I, Class II and Class III based on risk level, intended use,and regulatory requirements. This classification system is widely adopted in U.S. FDA regulations and closely aligns with the risk-based classification under the EU Medical Device Regulation (MDR). Both classifications directly impact injection moulding requirements.
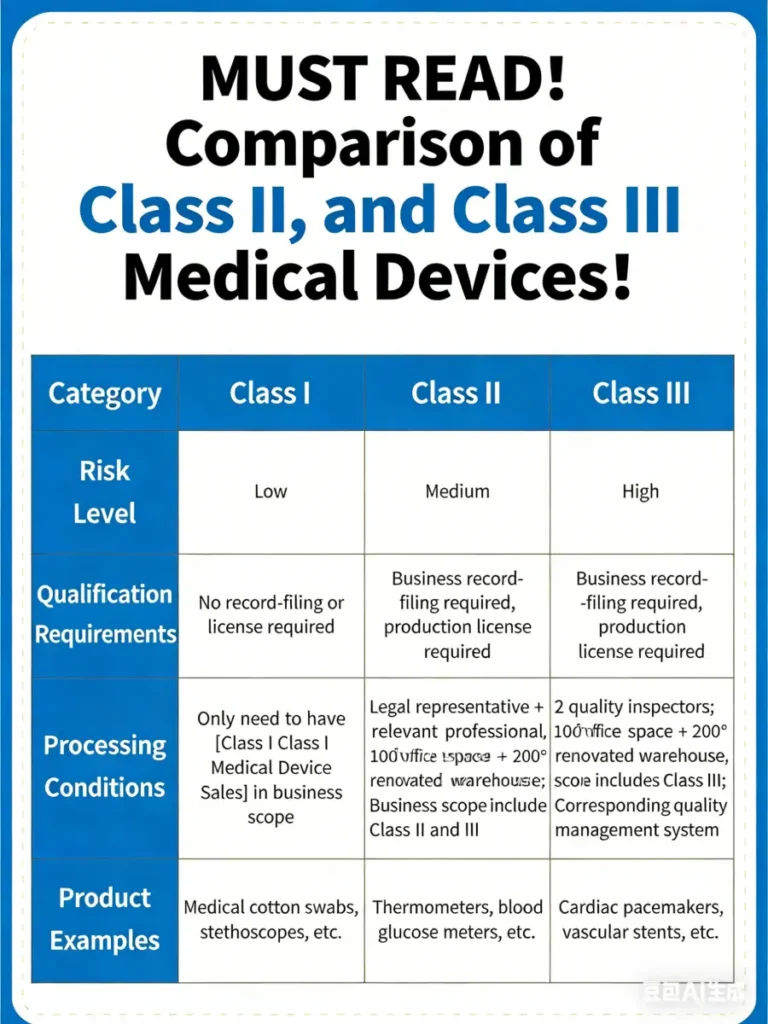
1. Class III High-Risk Devices
Materials: Must utilize medical-grade specialty plastics such as PEEK, with verified biocompatibility of material degradation products.
Molds: Employ S136 fully hardened stainless steel with cavity surface roughness below 0.1μm to prevent tissue irritation post-implantation.
Process: Deploy intelligent monitoring systems to control injection parameters in real time, achieving a product qualification rate exceeding 99%.
2. Class II Medium-Risk Devices
Materials: Must meet biocompatibility requirements, but modified engineering plastics may be selected.
Molds: Moderate precision tolerances of ±0.02mm are required, with optimized venting systems to prevent trapped air defects.
Process: Employ multi-stage injection moulding to control internal stresses and ensure product resistance to sterilization processes.
3. Class I Low-Risk Devices
Materials: Standard medical-grade plastics compliant with FDA or CE certification.
Molds: Utilize pre-hardened steel, prioritizing multi-cavity mold optimization for production efficiency.
Processing: Parameter control is relatively flexible, but batch quality consistency must be guaranteed.
What Is the Future Development of the Medical Injection Moulding Field?
As a core component of medical device manufacturing, the future development of the medical injection moulding field will revolve around four main areas: materials, technology, intelligent production, and product quality and safety. This will address the growing demand for personalized customization services, high precision, and safety compliance.
Materials
High-Performance Materials: Advanced materials like polyether ether ketone (PEEK) and polysulfone (PSU) will see broader adoption to meet stringent requirements for strength, durability, and biocompatibility in implants and surgical instruments.
Functional Materials: Research will prioritize antimicrobial plastics, chemically resistant materials, and biodegradable options to reduce post-operative infection risks and extend device longevity.
Technology
High Precision: injection moulding technology will pursue greater accuracy and the ability to form complex shapes to meet production demands for miniaturized, precision medical devices like minimally invasive surgical instruments.
3D Printing Technology: 3D printing will complement traditional injection moulding for producing customized medical devices, enhancing shaping precision and production flexibility.
Intelligent Manufacturing
To ensure product consistency, reduce defect rates, and boost production efficiency, manufacturers will widely adopt AI-assisted quality monitoring, automated inspection, and smart factory systems.
Product Quality And Safety
Stricter requirements will be imposed on production environments, material sterilization processes, and product traceability systems to ensure medical safety.
In summary, the future of medical injection moulding will be technology-driven, highly customized, intelligent, efficient, and safe. Companies must continuously invest in R&D, embrace intelligent transformation, and strictly adhere to global regulatory standards to maintain competitiveness in the evolving landscape.
Conclusion
Injection moulding technology is widely adopted in medical device manufacturing due to its capabilities for high-volume production, consistent quality, and high precision, coupled with the material’s excellent biocompatibility, sterility, and suitability for high-cleanliness environments. For manufacturers in the global medical device market, injection moulding offers a scalable, compliant, and cost-effective manufacturing solution.
Injection moulding will continue to meet the medical device industry’s evolving demands for precision, material stability, and controlled production environments. If you are considering selecting suitable materials and partnering with an experienced manufacturer, please contact us. We offer comprehensive services from mold design to finished product manufacturing.

Felix Lu
Senior DFM Expert · Huashuo Molding
Industry Exp.
Projects
DFM Optimization
Felix Lu has 16+ years of experience in mold manufacturing, DFM, and mass production, with a strong commitment to sharing advanced technologies and practical industry insights.